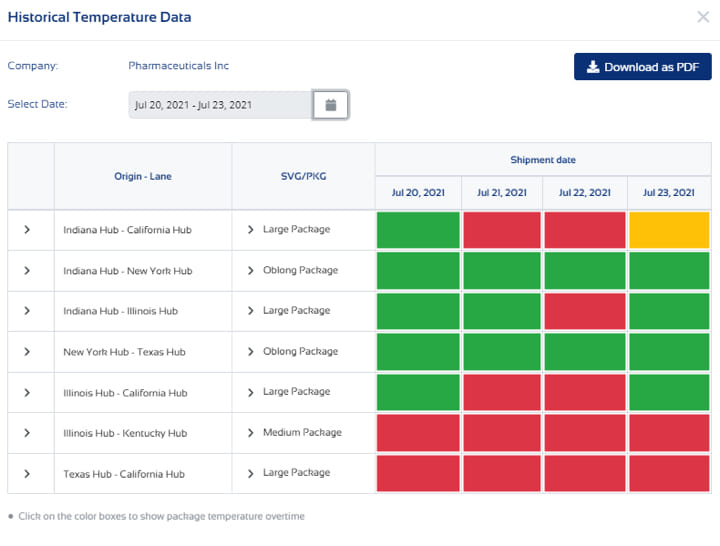
Radiopharmaceutical Logistics: Managing Decay, Timing, and Precision in Advanced Therapies
Radiopharmaceuticals operate under one defining constraint: time is a non-negotiable asset. Unlike traditional biopharmaceuticals,...
read DetailsCold Chain Engineering